Tag: water’
Outdoor Science for Kids: Water Rockets!
- by KitchenPantryScientist
This summer, get off screens and get outside with this fun project from my book Kitchen Science Lab for Kids!
Hot Weather Science
- by KitchenPantryScientist
Too hot to bike? Pool too crowed? Try some hot weather science! Just find a shady spot in the driveway, pull out the garden hose and you can experiment with:
siphons,
water rockets,
and fire-proof water balloons.

Click on the experiment names to read directions and learn more about the science. Have fun!
Simple Water Rockets
- by KitchenPantryScientist
You’ll have a blast learning physics by making water rockets!
While NASA’s rockets use rocket fuel as their working mass, these rockets use water. As pressurized air forces the water out of your rocket, the rocket moves in the opposite direction, just like Newton’s Third Law says it will. “For every action, there is an equal and opposite reaction.”
Although these rockets lack fins, a payload and a nose cone, you can see from this NASA illustration that they’re very similar to real rockets. You can make a complicated launch pad like this, but we decided to make things easy.
For this experiment, you’ll need:
-an empty one or two liter bottle from a carbonated beverage
-a cork that has been cut in half and will fit in the mouth of your bottle (An adult should do this. I used a serrated knife.)
-a needle for inflating balls
-a bike or ball pump
-a cardboard box cut to hold the bottle at an angle pointing away from you. This is your launch pad.
Push the needle through the cork so that it pokes out of the other side. Use the hole from the corkscrew to make it easier.
Fill the bottle about 1/3 of the way full of water and insert the cork in the bottle.
Set the bottle in the cardboard box so that it’s pointing up, but away from you. See the photograph.
Attach the needle to the bike pump, stand behind the launch pad and start pumping air into the bottle. The air pressure will build in the bubble at the top of the rocket. When the pressure gets high enough, it will force the cork and water out of the bottle with lots of force, and as the water shoots down, the rocket will shoot up!
What happens if you add more water, or less water to your rocket?
Can you imagine riding a real rocket? Check out Astronaut Abby’s website to meet a girl who wants to ride a rocket some day and ask an astronaut on the International Space Station questions!
Surface Tension in Space
- by KitchenPantryScientist
If you’ve done an experiment where you drip water onto a penny, or made Tie Dye Milk, you know what surface tension looks like here on Earth. How does it look in space?
Here’s an amazing video demonstrating how the surface tension of water looks in zero gravity on the international space station. Fascinating!
Survival Science: Water Filters
- by KitchenPantryScientist
 I demonstrated this experiment on Kare11. Click here to watch the clip.
I demonstrated this experiment on Kare11. Click here to watch the clip.
Don’t drink the water you purify when you do this experiment at home, since it may still contain harmful microbes!!! You can use homemade Petri dishes to see how clean you got your water though, by swabbing the unfiltered water on one dish and the water you purified on another to see what grows!
If you’re desperate for water, and you don’t have a plastic bag, or it’s too cloudy or cool to get water from tree respiration, you can always look for a pond or stream as a water source. Unfortunately, pond and stream water is often teeming with life, and some of the bacteria, viruses, parasites, protozoa and fungi in the water can make you sick. Boiling water purifies it, but what if you can’t make a fire? Katniss had Iodine in her backpack, which is another great way to kill microorganisms and purify water, but what if you don’t have iodine or a water filter?
If you’re in a survival situation, it’s important to do everything you can to make sure the water is safe, before you drink it! The first thing you have to do is make the water you find clear enough to read a paper through (get out the gunk, in other words.) Then, you have to put it in the sun for six hours so ultraviolet rays will kill anything living in the water. Digging a hole next to a pond and collecting the water that seeps in will pre-filter the water for you!

Using grass, charcoal, sandy soil, you can make a water filter. A plastic water bottle with the bottom cut off works best as a filter-holder, but you can use a cone made of birch bark if you don’t have a bottle!
We cut the bottoms off of some water bottles, pounded up some charcoal (you could use campfire coals for the charcoal layer!), pulled some grass and found some sand (don’t use sand from a sandbox, which may contain cat feces.)
Next, we layered grass, charcoal and sand in our bottles, packing each layer down as much as possible. I tried grass, charcoal, sand, charcoal and more grass, but the girls made more layers than I did. We wrote down what we did to see who got the cleanest water!

Then, we poured stream water through our filters and collected what flowed through in empty sports drink bottles. We covered the bottles and left them in the sun for at least six hours. Last night, we stuck cotton swabs in each water sample and swabbed homemade petri dishes to test the water for fungi and bacterial growth. (You can’t see viruses, but the sun will often kill them efficiently.) Our controls were stream water that was unfiltered and keep out of the sun and stream water that was unfiltered, but put in the sun with the other samples. Note: If you want to make petri dishes to test your water, use agar instead of gelatin to make the plates! Some bacteria are good at breaking down gelatin, turning it into a liquid, and this happened to a few of our samples (including the unfiltered water.)

We observed that putting the water through our filters made it yellow and one sample was a little cloudy, so it will be interesting to see how it compares to the stream water we didn’t filter or put in the sun. It would be fun to test the filter ingredients to see what made it yellow.
Survival Science: Water Collection
- by KitchenPantryScientist
At the beginning of “The Hunger Games,” by Suzanne Collins, the heroine Katniss risks her life to retrieve an orange backpack and a sheet of plastic. In the backpack, she finds a black sleeping bag, a pack of crackers, a pair of dried beef strips, a bottle of iodine, a box of wooden matches, a small coil of wire, a pair of night glasses, a bottle of iodine and a half-gallon plastic bottle for carrying water. According to the book “Stay Alive,” by John D. McCann, she’s off to a pretty good start in terms of basic survival. He says that the following components should be included in any survival kit: fire and light, signalling, water and food collection, knives and tools, shelter and protection, navigation, and medical. I demonstrated how to do this experiment on Kare11. Click here to watch the clip.
The survival “Rule of Three” says that you can go 3 minutes without air, 3 hours without shelter (from heat or cold), 3 days without water and 3 weeks without food. That means that water is one of the first things you have to think about in a survival situation. Your body is about 75% water.
If it’s hot outside, you can easily collect water from non-poisonous leafy trees using a plastic bag. Clear works best, like a large oven bag. (Ours was for turkeys!)
Yesterday, we took 2 bags outside and put them over tree branches, enclosing as many maple leaves as we could and putting a small rock in the corner of each bag to weight them. Finally, we tied them around each branch securely with a twisty-tie. What else could you use if you didn’t have a tie? A shoelace? After 24 hours, we collected our water by cutting the corner of the bag and collecting it in a clear bottle.
Katniss had iodine to purify her water, but if you don’t have a water filter or iodine tablets to kill harmful bacteria, viruses and parasites, you could filter the clear water from plant transpiration through cloth, like a tee shirt or bandana, and leave it in a clear bottle in the sun for 6-10 hours to kill most pathogens. Don’t try drinking the water you get from this experiment, as it may not be safe!
Why to trees produce water? All plants carry water from their roots to small pores called stromata on the underside of their leaves. These pores release water into the air in a process called transpiration. Transpiration helps cool plants, but is also involved in moving nutrients from root to leaf. Plants transpire the most on hot, dry days and trees that have gotten plenty of water give off the most water. According to the U.S. Geological Survey, a large Oak tree can transpire 40,000 gallons of water a year! Our area is very dry at the moment. We probably could have collected more water from our tree following some rain!
So, as of today, our survival kit will include a pocket knife, a clear plastic oven bag, and a bandana. What should we add next?
Solar Water Purification
- by KitchenPantryScientist
How would you get water to drink if you were stranded on a desert island, surrounded by salty ocean? If you happened to have a big leaf and a few concave (bowl-like) items- maybe a big shell and a smaller shell, you could make a solar water purifier to collect drinking water.

Even if you’re not on a desert island, it’s fun to make your own solar water-purifier using a big big bowl, a small bowl, some plastic wrap and a marble or a pebble. You’ll also need salt and food coloring to “contaminate” the water you’ll be purifying. This experiment works best on a hot, sunny day since it harnesses the power of the sun’s rays to clean the water.
Put the small bowl inside the big bowl. The top of the small bowl must be lower than the top of the big bowl.
In a separate container, mix together water, salt and food coloring until you have a mixture too salty to drink. Pour the water into the big bowl, making sure the liquid stays outside of your small bowl, since you’ll be collecting clean water in the small bowl.
Loosely cover the top of the bowl with a single piece of plastic wrap. Place a pebble or marble in the center of the plastic wrap and adjust the wrap so that there is a dip directly above the small collection bowl. Try to seal the plastic wrap around the edges of the bowl as well as you can.
Now, place the bowl in the sunlight. The sun’s ultraviolet rays will go through the plastic wrap and into the colored water, where they’ll be absorbed and re-released as heat energy. Since the heat can’t escape back out through the plastic wrap, the air and water in the bowl heat up.
The warm temperature helps water molecules on the surface evaporate, or escape, into the air in the bowl. When they collide with the plastic wrap, they encounter a cooler surface since the air outside the bowl is not as warm. This causes them to condense, or form droplets, on the plastic wrap. When the droplets get big enough, gravity pulls them to the lowest part of the plastic wrap (the dip) and they drip into the collection container.
When you’ve collected enough purified water (be patient- it can take a day or two), you can taste the water to see how your purifier worked. Be sure to wipe of the bottom of the collection bowl before you pour it out so you don’t contaminate your clean water!
A variation on the experiment is to add vinegar to the water, purify it, and check the pH of your starting and purified water using litmus paper. You can make your own litmus paper using red cabbage!
Be Thankful for Clean Water
- by KitchenPantryScientist
This Thanksgiving, here is what I’m thankful for:
A nasty stomach virus burned through our house this week (I blame a communal bowl of popcorn.) Following the kids around, I cleaned up after them with disinfectant wipes and lots of soap and water. If I didn’t hear the comforting swoosh of the faucet following the sound of a flush, I’d yell “wash your hands!” When they emerged from the bathroom, I’d hand them a glass of water or bottle of sports drink to sip so they could replace the fluids they’d lost.
In Haiti, for most people there are no clean toilets and there is no clean water for hand washing. There often isn’t clean drinking water and ditches are filled with sewage. Over 1,000 people have already died in a recent cholera epidemic and countless more are infected. Cholera is a disease of people living with poor sanitation.
What can you do to help?
Click the link above to donate a day of clean water to someone in the third world. Here’s what P&G is doing now to help with the cholera epidemic in Haiti. You can click once a day!
According to the World Health Organization, waterborne diseases remain the leading cause of illness and death in the developing world. Every 20 seconds, a child in a developing country dies of a water-related illness. Bacterial diseases caused by E.coli and Vibrio Cholerea, parasites like Guinea Worm, viruses and protozoa are all carried by unclean water. Some of these potentially deadly organisms can be killed by boiling, but for many people in the world, firewood is scarce.
We don’t know how lucky we are. Be thankful for your clean drinking water, even if it tastes like chlorine. Be thankful for your toilet.
Happy Thanksgiving and don’t forget to wash your hands!
Many aid organizations and churches have programs to help people get access to clean water and proper sanitation, whether it’s by helping dig wells or sending bars of soap. WaterAid is a U.K.-based international non-profit organization that is helping bring not only clean water, but sanitation and hygiene programs to many villages desperate for clean water. The organization makes local women an integral part of the process. Proctor and Gamble, who is running the Clean Water Blogivation campaign, make a PUR powder which can be mixed with contaminated water to make it safe to drink. They have a Children’s Safe Drinking Water program and have been working with a number of organizations to distribute the PUR powder around the world to those who don’t have access to clean water.
There’s a fairly comprehensive list of water-born microbes on Wikipedia, and if you’d like to learn more, the World Health Organization and the Center for Disease Control are great resources.
What We Take for Granted
- by KitchenPantryScientist
The facts speak for themselves:
- Every 20 seconds, a child in a developing country dies of a water-related illness (World Health Organization)
- Waterborne diseases remain the leading cause of illness and death in the developing world. (World Health Organization)
- 46 percent of people on Earth do not have water piped to their homes (National Geographic)
- Women in developing countries walk an average of 3.7 miles to get water (National Geographic)
Shocking, isn’t it? It should at least give you pause as you fill up your child’s glass with clean tap water, or maybe even filtered tap water. Now, try to imaging walking 3.7 miles in the dark or the hot sun to bring home water that may or may not make your child sick, or even kill them. What choice would you have though? Water is life.
I recently signed up to participate in the Clean Water Blogivation campaign. If my blog receives the most votes, I could win an opportunity to be a change agent and join Dr. Greg Allgood on a clean water expedition to Africa and a $15,000 donation to my favorite charity tackling water issues. Much more importantly, each time someone votes for my blog post, P&G will donate a day’s worth of clean drinking water (2L) to a person in need in a developing country. Click on the “vote for this blogger” button above and clean drinking water for a day will be donated each time you vote! (You can vote once a day.)
The task of fetching water defines life for many people on this planet. National Geographic tells the story of an Ethiopian woman named Aylito who dropped out of school when she was eight to help her mother carry water from a dirty river. She spends eight hours a day walking to and from the river, up and down a mountain, three times to carry 50 pounds of water on her back. It is a life almost beyond imagining for most of us who live in developed countries. However, one can imagine that people who work so hard to get so little water have very little water to spare for sanitation, like hand-washing, or washing clothes. According to National Geographic, proper hand washing alone can reduce diarrheal disease by 45%. Many people cannot afford soap though.
What diseases are carried by dirty water? You name it: bacterial diseases like E.coli and Vibrio Cholerea, parasites like Guinea Worm, viruses and protozoa. Some of these pathogens can be killed by boiling, but for many, firewood is scarce. Sometimes, it’s too much water from flooding that causes drinking water to be unsafe. (The floods in Pakistan are causing problems right now.)
There’s a fairly comprehensive list of water-born microbes on Wikipedia, and if you’d like to learn more, the World Health Organization and the Center for Disease Control are great resources.
The obvious question is, “what can I do to help?” Many aid organizations and churches have programs to help people get access to clean water and proper sanitation, whether it’s by helping dig wells or sending bars of soap. WaterAid is a U.K.-based international non-profit organization that is helping bring not only clean water, but sanitation and hygiene programs to many villages desperate for clean water. The organization makes local women an integral part of the process. Proctor and Gamble, who is running the Clean Water Blogivation campaign, make a PUR powder which can be mixed with contaminated water to make it safe to drink. They have a Children’s Safe Drinking Water program and have been working with a number of organizations to distribute the PUR powder around the world to those who don’t have access to clean water.
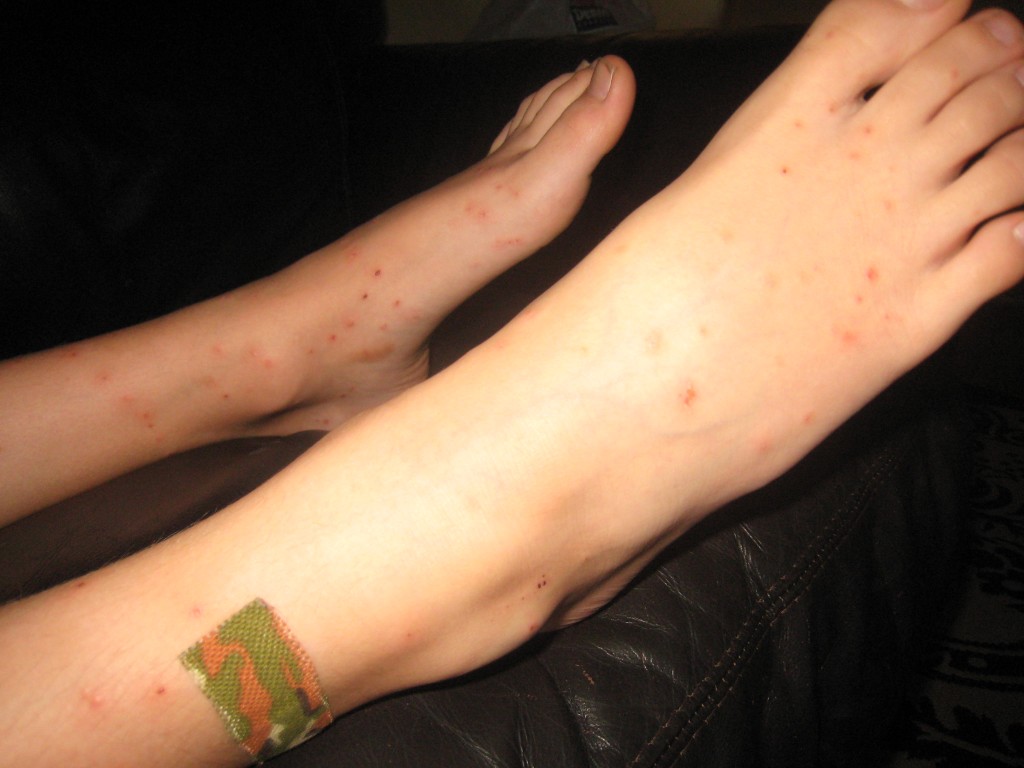
Beware the Pretty Birds!
- by KitchenPantryScientist
We spent last week in Iowa at a family reunion at lake Okoboji, a deep, clear, gorgeous lake near the Minnesota border. My great grandparents had a cottage there, and every year my mom’s side of the family returns to spend a week swimming, fishing, going to the lakeside amusement park and just hanging out.
Gull Point is a pretty piece of land with a sandy beach and sandbar that juts out into the lake and is one of our favorite destinations. Each year we spend an afternoon or two there, swimming, skipping rocks and digging our toes into the sand. We didn’t see many gulls this year, but there was a pretty white pelican hanging out nearby. Little did I know that water birds carry a tiny parasite that infects snails in the water. The parasite’s larvae are then released and swim around looking for a suitable mammalian host to burrow into and continue their life cycle.
Ignorant of the tiny bugs digging into our skin, we frolicked in the water for hours.
Humans are not a good host for these parasites and they soon die, leaving their buggy corpses under our skin. Some people don’t have much of a reaction, but others have immunes systems that kick into high gear, rejecting the dead parasites with an inflammatory response in the the form of big, itchy, awful bumps.
My son is definitely allergic. The CDC website has more information on Swimmer’s Itch, or Cercarial Dermatitis, which can also be found in many Minnesota lakes. Generally, it doesn’t need much treatment except Hydrocortisone cream or Calamine lotion. My daughter and I have a few itchy bumps, but nothing major. My son, on the other hand is on oral prednisone too calm his allergic reaction. Needless to say, it has been a long, long week.
According to the University of Minnesota’s Extension Service, you can do the following things to reduce your chances of getting severe swimmer’s itch:
- Dry off as soon as you leave the water. Rub skin briskly to remove water drops before they evaporate. Be sure to dry underneath waistbands and around leg openings. Encourage children to dry off thoroughly each time they leave the water.
- Shower with soap and fresh water or change into dry clothes as soon as possible.
- Don’t wade or play in shallow water. Swimming from a raft or pontoon minimizes your exposure.
- Don’t feed geese and ducks near your beach. Waterfowl are an important adult host for the parasites.
You can also go to http://checkmylake.com to search for specific lakes to see if they’re safe for your family to swim in. I wish I would have checked!